Influenza has a long history of being one of the most deadly diseases to afflict humanity, but what exactly makes it so dangerous?
Certainly one reason is that influenza viruses have a history of jumping from other animals to humans, which, when the trans-species virus is new to the human population, generally means that human immune systems have no natural resistance. Another reason is that influenza viruses, with their rapidly mutating single-strand RNA genomes, are highly variable over time.
Now a more complex and daunting view is emerging that shows viruses' constant variability -- and sometimes deadly innovation -- happens as a result of these two factors, combined with influenza's tendency to swap genes among viruses and also to move between geographically separate populations through traveling or migrating hosts.
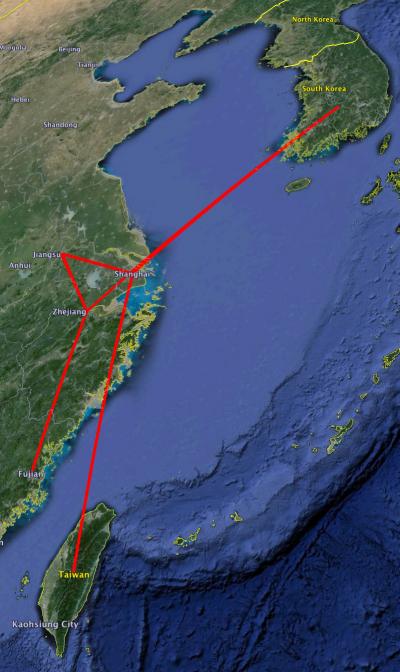
routemap of the earliest isolates of H7N9 indicating that the HA gene came in from Korea to Shanghai and Zhejiang and then spread. Credit: Daniel Janies
A new report published online on January 21, 2015 in the journal Cladistics sees H7 influenza not just as a steadily changing virus, but as regularly shuffling combinations of genetic segments, each of which has its own history of travel among hosts and geographic places.
A host can often be infected with more than one type of virus and, as viruses replicate in the host's cells, the genetic segments of the progeny viruses can be shuffled into new combinations. In essence, the influenza viruses are made of set of generally interchangeable parts that periodically assemble a virus newly suited to evade hosts' immune systems.
"A case in point is the China-Taiwan H7N9 outbreak, which was caused by a virus made of genetic segments previously circulating in chickens in China and ducks in Korea," the authors note. "Some segments have historical international connections to and from China-Taiwan to neighboring countries."
Appearing in the journal's February 2015 issue, the study, "Phylogenetic visualization of the spread of H7 influenza A viruses" uses genomic analysis to look at the phylogenetic history of genes that assort with H7 influenza. H7 viruses infect a variety of birds and periodically infect humans including the recent human cases of H7N9 in China, Hong Kong, and Taiwan. The paper's authors are University of North Carolina at Charlotte's Daniel A. Janies and Chris Krueger, and Laura W. Pomeroy, Igor O. Voronkin, Jori Hardman, Yuqi Zhang, Izzet Senturk, Kamer Kaya and Ümit Çatalyürek from the Ohio State University.
Contrary to traditional opinion that H7 is a restricted in geographic scope, the authors show regular transit in genes that assort with H7 viruses across vast geographic distances. "We find multiple occurrences of transboundary and transcontinental spread, both within and between the previously observed American and Eurasian clusters," the authors say.
"H7 strains have a long history," said UNC Charlotte's Janies. "There are isolates of H7 going back to 1902. But the recent H7N9 virus is a new constellation of genes - a new disease, based not only on H7, but other strains of influenza"
The view that that individual influenza genes have moved between species and across great geographic distances, complicates the issue of viral adaptation and evolution. Concurrent with reassortment of genes into new sets, a specific gene's mutation rate can be rapid, and thus the possibilities for dangerous new viral adaptations are constant.
Despite the dynamic nature of the virus, most research labs construct phylogenetic trees gene-by-gene. However these trees are difficult to interpret. Janies and colleagues have developed new computing techniques that combine viral genomic data and geographic metadata to create visualizations of viral traffic across the planet (http://geogenes.org). The results, called transmission graphs, allow public health scientists to identify hubs for the transport of the disease where countermeasures will have the most durable effect on the global spread of the disease.
Janies argues a new approach is needed in how we study and monitor widespread zoonotic viruses like influenza that frequently cross species and geographic boundaries. While the global health community jumps into action when outbreaks in humans occur, if the outbreak is stopped it does not mean that the danger has fully subsided because the reservoirs for the viruses may be distributed elsewhere among animals and geography.
"There's an unlearned lesson: even if people say the disease went away, often it hasn't - the virus can be hanging around in places and hosts at which we are not looking, it's hanging around in chickens, it's hanging around in pigeons - our tools enable researchers to know where to focus efforts for the best chance to break the transmission network." Janies said, noting that the wildlife biologists and veterinary doctors who know and observe animal diseases often do not often belong to the same communication networks as physicians.
"The viruses don't care what discipline in which you work. You are going to have patients and problems caused by the viruses crossing over geographic boundaries, host boundaries and disciplinary boundaries," he said.
"When a host is co-infected, the genes assemble themselves into novel viruses to which people may not be immune. It's not just that the genes mutate, it's that the whole deck gets shuffled. That's what happened with H7N9 recently and pandemic H1N1 from 2009. New combinations are important things for which to watch, no matter where they occur."




Comments