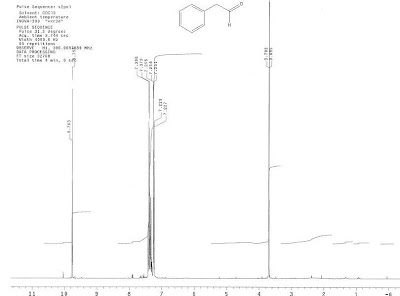
Although the HMR of 37-F1 shows reasonable purity for use as a reagent, it appears contaminated with droplets floating and sticking to the sides of the flask. This may be water condensed by the distillation in open air. Future distillation should be done under nitrogen. Phenylacetaldehyde is reported to boil at 195C. Since 37-F1 was collected at 162C (approx), perhaps this corresponds to an azeotrope with water and would explain the contamination in the first fraction. In the future more careful measurement of the Ubergang temperature over the course of the distillation would help to clarify this issue.4) The history of this wiki page reveals who made each contribution. A major advantage of using a hosted wiki as a lab notebook is that a reference to every version of the notebook can be made, marked with a third party time stamp: On October 27, 2006 James recorded his log, the NMR results and a conclusion. On October 29, 2006 I added some questions in bold, re-interpreted the results, added a detailed discussion and changed the conclusion. On January 3, 2007 James corrected a spelling mistake I made. On January 9, 2007 my graduate student Khalid added a link for phenylacetaldehyde. On January 23, 2007 Khalid added InChI tags. On November 22, 2007 James answered my questions in the log. 5) This is the kind of information that someone experiencing problems with purifying phenylacetaldehyde could use. In the experimental section of a chemistry article the most you are likely to get is "phenylacetaldehyde was purified by distillation", with no proof as to the actual purity. Notice that the experiment is never really "completed". There is always more that someone could add in terms of the discussion or add a question anywhere in the document. That is how science actually happens for the most part and we can forget that when listening to a slick presentation at a conference or read a well-written article making it look like the project unfolded effortlessly from a perfect plan. Coming back to the person who searched for and found this page. This lab notebook page may or may not help solve their problem directly. The most valuable information is probably the contact information for the people who contributed to the execution and analysis of the experiment. We aren't hiding behind pseudonyms. We use our real names because we want to fully participate with the research community and we stand behind what we report. Shoot us an email for help if the notebook isn't enough.
How Should Open Notebook Science Be Used?
Maxine Clarke highlights a bit of recent controversy regarding Open Notebook Science that has been bouncing around the blogosphere and FriendFeedosphere.
There are some who interpret the ongoing publication of our laboratory notebook as an expectation for the world to read it like a magazine. For someone who is not a collaborator or working in a related area that would make about as much sense as reading the phone book.
Here is an example of how an Open Notebook should be used:
Based on our Sitemeter records, this morning someone from the UK seached for "purification of phenylacetaldehyde" on Google. The first hit was the UsefulChem experiment EXP037 performed by undergrad student James Giammarco on October 6, 2006.
This happens to be a "failed experiment". But here is the information that can be obtained from the wiki:
1) The picture of the experimental set up:
 2) The results provide an NMR of the first distillate, showing exactly the nature and amount of impurities. This is before we started using JSpecView systematically so this spectrum is not interactive, it is just an image.
2) The results provide an NMR of the first distillate, showing exactly the nature and amount of impurities. This is before we started using JSpecView systematically so this spectrum is not interactive, it is just an image.
 3) The discussion addresses a major problem with the first distillate and a suggestion is made about what might be the problem:
3) The discussion addresses a major problem with the first distillate and a suggestion is made about what might be the problem:





Comments